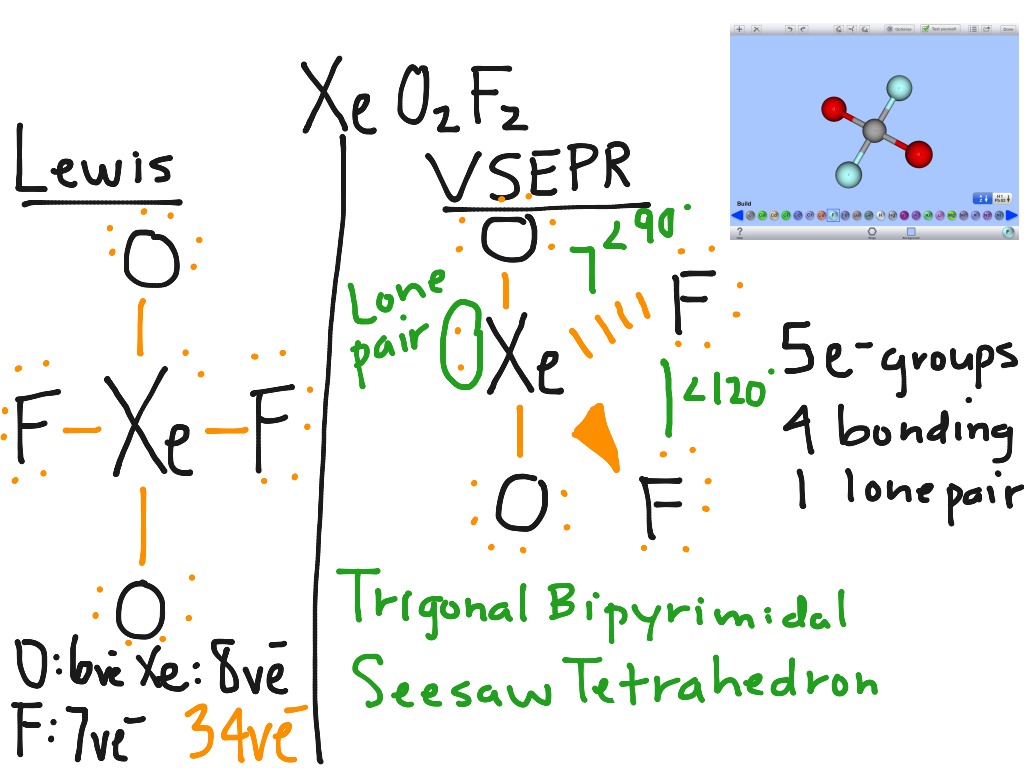
The molecular geometry of SO2 is bent or V-shaped, with the sulfur atom at the center and the oxygen atoms on either side. Each oxygen atom is connected to the sulfur atom by a single bond, and there is a lone pair of electrons on the sulfur atom. In the Lewis structure, the sulfur atom is in the center, surrounded by two oxygen atoms. The Lewis structure of SO2 can help us visualize the bonding arrangement. This charge distribution creates a dipole moment in the molecule, where one end has a slightly positive charge and the other end has a slightly negative charge. On the other hand, sulfur has a partial positive charge due to the unequal sharing of electrons. Oxygen’s higher electronegativity means it has a greater pull on the shared electrons, resulting in a partial negative charge on the oxygen atoms. Electronegativity is the ability of an atom to attract electrons towards itself in a chemical bond. In SO2, the oxygen atoms are more electronegative than sulfur. However, in a polar covalent bond, the sharing of electrons is unequal, resulting in a partial positive charge on one atom and a partial negative charge on the other. A covalent bond is formed when two atoms share electrons. In the case of SO2, the bond between sulfur and oxygen is a polar covalent bond. Explanation of the Type of Bond in SO2 (Polar Covalent Bond)  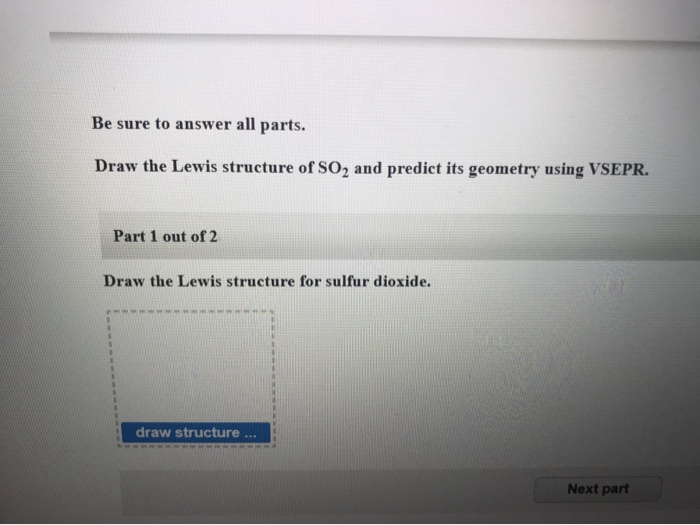
Valence electrons are the electrons in the outermost energy level of an atom that participate in bonding. In the case of SO2, sulfur has six valence electrons, while oxygen has six valence electrons each. To understand the bond type in SO2, we need to delve into the concept of chemical bonding.Ĭhemical bonding occurs when atoms share or transfer electrons to achieve a stable electron configuration. Sulfur dioxide (SO2) is a chemical compound composed of one sulfur atom bonded with two oxygen atoms. The polarity of SO2 allows it to dissolve in polar solvents and exhibit dipole-dipole interactions.The difference in electronegativity between sulfur and oxygen atoms creates a dipole moment, making SO2 polar.The molecule has a bent shape due to the presence of two lone pairs of electrons on the sulfur atom.SO2 (sulfur dioxide) is a polar molecule.Keywords: sulfur dioxide, chemical compound, polar, nonpolar, molecule, electrons, shape, bent, V-shaped, sulfur atom, oxygen atoms, electronegativity. To understand why SO2 is polar, we need to delve deeper into its molecular structure and the concept of electronegativity. This bent shape, along with the electronegativity difference between sulfur and oxygen, leads to the molecule being polar. In the case of SO2, the molecule is bent or V-shaped, with the sulfur atom at the center and the two oxygen atoms on either side. The polarity of a molecule is determined by the distribution of its electrons and the shape of the molecule. One of the key questions surrounding SO2 is whether it is a polar or nonpolar molecule. It is commonly found in volcanic gases, industrial emissions, and as a byproduct of certain chemical reactions. Sulfur dioxide (SO2) is a chemical compound composed of one sulfur atom and two oxygen atoms.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
